Programs
Expanded Access Programs (EAPs), also commonly referred to as compassionate use, refers to the use of an investigational therapy outside a clinical trial when the primary purpose is to diagnose, monitor, or treat a serious condition in a patient. This is different from a clinical trial, where more comprehensive information on the safety and effectiveness of an investigational therapy is collected.
Sometimes it isn’t possible to take part in a clinical trial. If that’s the case, your doctor may be able to request access to an investigational therapy through an Expanded Access Program.
Through an EAP, a doctor can request an investigational therapy for someone if their current therapy isn’t working or if they have no other treatment options. These programs are closely monitored by the participating doctor to make sure they are conducted in a manner that puts the safety of the participant first.
The primary purpose of an EAP is to provide access to an investigational therapy outside a clinical trial if:
- The patient cannot participate in a clinical trial because the trial is no longer enrolling / open or they are not eligible
- The patient meets the EAP requirements
- No other comparable therapy options are available or working
- The patient and their doctor agree that the EAP is the best potential option for treatment of their disease
The safety and well-being of patients being treated through the EAP remains the top priority, and patients may choose to stop participating at any time.
Participating in an EAP varies from program to program but in general you can expect the following:4,5
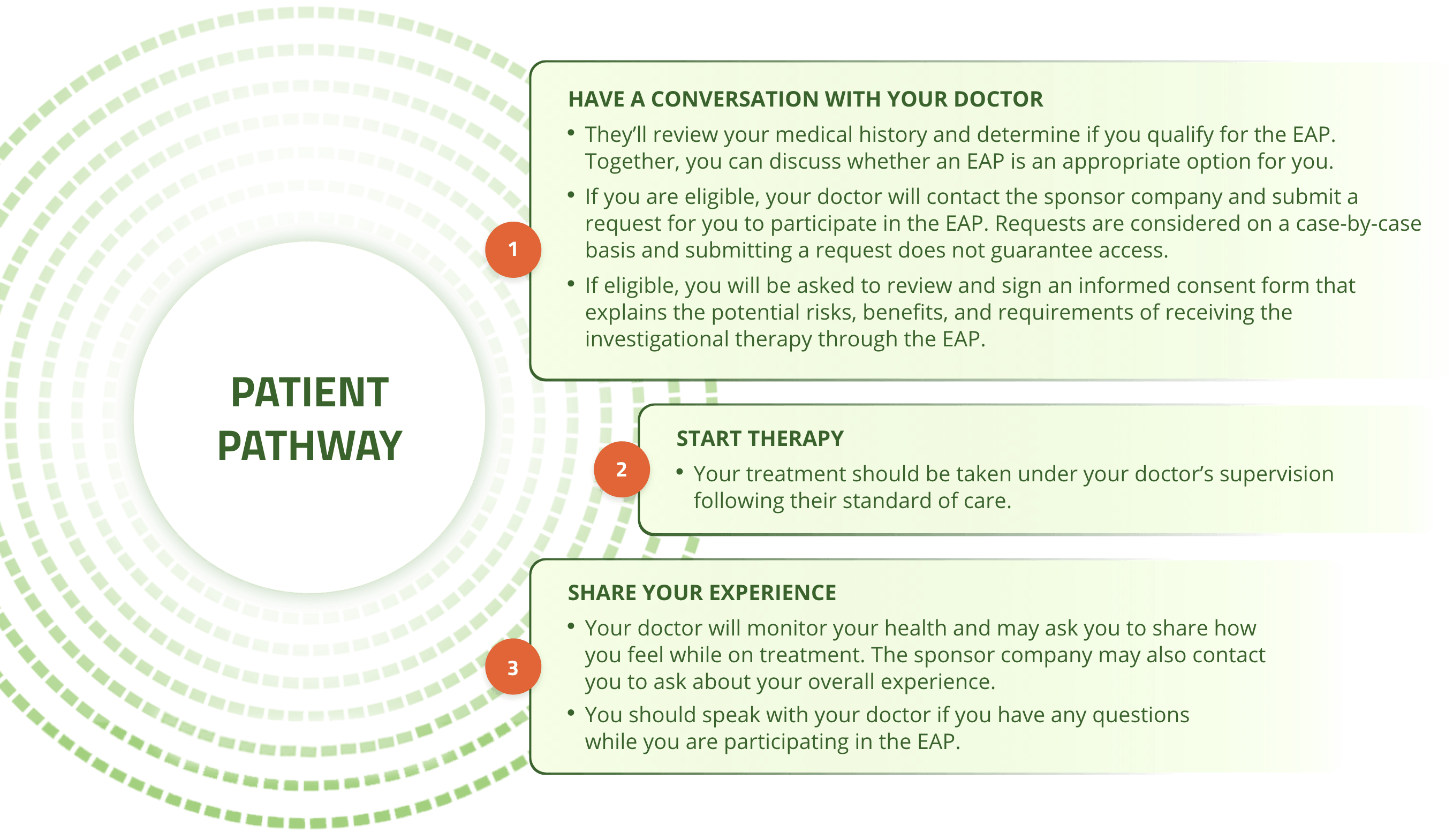
Cogent Biosciences has initiated an EAP with the investigational therapy bezuclastinib for patients with systemic mastocytosis (SM) or gastrointestinal stromal tumors (GIST) who meet disease-specific protocol inclusion and exclusion criteria.5
To determine if participating in an EAP is the right option for you, your doctor will evaluate you and decide if you qualify for early access to bezuclastinib. They will discuss the potential benefits and risks of the EAP and bezuclastinib, and together you will decide if requesting to participate in the program is the right option for you.
References
- Expanded Access, Information for Patients. U.S. Food and Drug Administration. Updated November 7 2022. Accessed January 15 2026. https://www.fda.gov/news-events/expanded-access/expanded-access-information-patients#process-work
- Our Expanded Access Policy. Cogent Biosciences. Accessed January 15 2026. https://www.cogentbio.com/bezuclastinib-program-development/#our-expanded-access-policy



