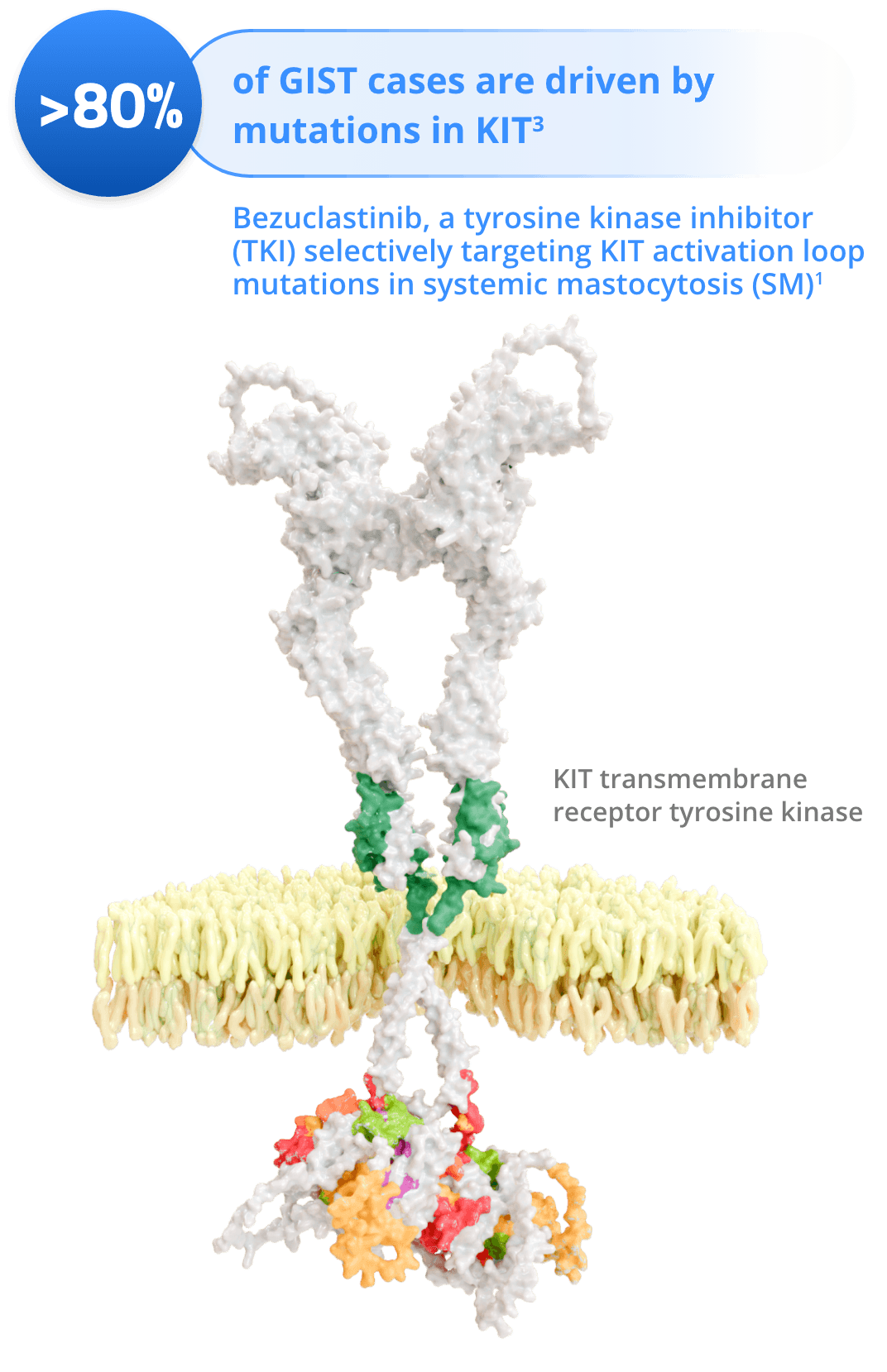
Cogent’s lead drug candidate, bezuclastinib, is designed to target exon 17 mutations found within the KIT receptor tyrosine kinase, including KIT D816V. When KIT D816V remains in a perpetual ‘on’ state causing mast cells, a type of white blood cell, to accumulate in various internal organs including the bone marrow. The result is an orphan disease called Systemic Mastocytosis (SM).
Exon 17 mutations have also been found in advanced Gastrointestinal Stromal Tumors (GIST), which have a strong dependence on oncogenic KIT signaling. Bezuclastinib is a highly selective and potent KIT inhibitor with the potential to provide a powerful new treatment option for patients with both of these diseases.
OUR APPROACH
Cogent is focused on creating best-in-class potent and selective small molecule inhibitors for patients in need of better treatment options. Our broad portfolio of novel agents demonstrates our ability to discover and advance targeted therapies in both hematology and oncology.
Choose an area below to learn more:
Systemic Mastocytosis occurs when mast cells inappropriately accumulate in various internal organs in the body. About 90% of people with Systemic Mastocytosis have Nonadvanced Systemic Mastocytosis (NonAdvSM), a life-long illness with chronic symptoms including headaches, urticaria pigmentosa, skin lesions, skin redness and warmth (flushing), abdominal pain, bloating, vomiting, diarrhea, and gastroesophageal reflux (GERD), that significantly impact the patient’s quality of life.
Many patients are also at high risk for severe, life-threatening anaphylactic reactions to various triggers such as insect bites. Advanced Systemic Mastocytosis (AdvSM) is a rare, very aggressive form of Systemic Mastocytosis. Patients with AdvSM may suffer from a multitude of debilitating symptoms such as anemia, thrombocytopenia, ascites, bone fractures, gastrointestinal abnormalities, and enlargement of the liver, spleen, and lymph nodes, which ultimately lead to organ failure and early death. Historically, the median life expectancy for AdvSM was less than 3.5 years.

Gastrointestinal Stromal Tumors (GIST) are categorized by uncontrolled cell growth in the tissues of the gastrointestinal (GI) tract. At diagnosis, about 80% of GIST patients’ tumors are the result of primary KIT mutations. Approved therapies currently inhibit a subset of these mutations, but most patients develop resistance due to additional secondary KIT mutations, including mutations in Exon 17. Bezuclastinib is designed to be a potent and selective inhibitor of KIT Exon 17 mutations, and by combining bezuclastinib with drugs that inhibit additional KIT mutations, Cogent is addressing a clear unmet medical need for patients with GIST.

We are pursuing the development of bezuclastinib in patients living with Systemic Mastocytosis and Gastrointestinal Stromal Tumors as patients wait for more effective medicines. Successful clinical trials are required to gain regulatory approval for new medications to advance patient care. If you are interested in participating or learning more about Cogent’s trials currently underway, please contact us.



Our Expanded Access Policy
Cogent Biosciences, Inc. (“Cogent”) is a company of people who are passionate and dedicated to turning the promises of our therapies into reality to transform the lives of patients. To accomplish this, we work with researchers, clinicians, patient advocacy groups, patients, and caregivers throughout the clinical development process. We believe that the most appropriate way to deliver on our commitment to patients is through completion of our clinical trials and subsequent successful registration of our medicine(s) as this will provide us with the greatest opportunity to potentially provide long-term access to as many patients as possible. Expanded access refers to the use of an investigational therapy outside a clinical trial when the primary purpose is to diagnose, monitor, or treat a serious condition in a patient. This is different from a clinical trial, where more comprehensive information on the safety and effectiveness of an investigational therapy is collected.
Cogent has initiated Bezuclastinib Expanded Access Protocols (EAP) for patients with GIST or SM who meet disease-specific protocol inclusion and exclusion criteria.
Expanded access will be considered when the following criteria are met:
- The patient has a serious or immediately life-threatening disease or condition.
- There is no comparable or satisfactory alternative therapy to treat the disease or condition.
- Potential patient benefit justifies the potential risks of treatment.
- Providing investigational therapy will not interfere with development or marketing approval for the treatment indication.
- The patient cannot have previously participated in, or cannot be currently participating in, a bezuclastinib clinical trial.
- Participation in an actively enrolling bezuclastinib clinical trial is not possible.
- Adequate supply of investigational therapy is available.
- The patient must reside in the United States.
How to Submit a Request for Bezuclastinib Expanded Access
If you are a patient, family member, or caregiver and want to know more about the Bezuclastinib EAP, please discuss this EAP, and all treatment options, with your treating physician. Requests for expanded access must be submitted voluntarily by the patient’s treating physician. Requests cannot be made directly by an individual patient or a patient’s parent/legal guardian or caregiver. Requests are considered on a case-by-case basis. Submitting a request does not guarantee access will be granted.
To initiate the process for possible participation in the Bezuclastinib EAP, licensed physicians, or a licensed physician’s staff member, should send an email to EAP@cogentbio.com. A response regarding the process for accessing the Bezuclastinib EAP will be provided within 2 to 3 business days of the initial request.
Physicians who obtain access to bezuclastinib through this EAP will be required to comply with all applicable laws, regulations, and contractual conditions, including, but not limited to, obtaining patient consent, patient monitoring and safety reporting, and obtaining appropriate institutional review board approvals.
Consistent with the 21st Century Cures Act, Cogent may revise this policy at any time.
Contact Us
For general inquires related to expanded access to medicines and our clinical trials, please contact the Cogent Medical Affairs Team at EAP@cogentbio.com.








